Dear Reader, you have faithfully followed us now into this 11th of our series of article on maintaining brain health as we age. Some of you have written to say "Enough, already! I've learned all about the brain. What supplements do I take?" As you'll discover next month, supplements are just a part of the program, but in the interest of helping those folks, and the rest of you, maintain a healthy blood pressure I will no longer delay my opinion.
For the dénouement, in the next newsletter we'll give final supporting information, including
- a brief economic analysis of these recommendations;
- brand names;
- lifestyle recommendations
- a list of other medications and supplements that reduce brain aging, and why they didn't make the cut;
- an analysis of medications that slow dementia as a side effect.
To allow me to wrap up all the loose ends of this long series, the March newsletter will be our longest newsletter to date. You can review previous articles at our website.
Test Your Current Brain Health
Section titled “Test Your Current Brain Health”After reading all the research and recommendations of this series on brain health, you may still be asking yourself what makes economic sense. Does the money you might shell out for supplements pay off in the long run?
We human beings hate getting sick and hurt, but for most of us the greatest horror is the loss of our ability to think, to maintain our competence in front of those we love and respect, and to take care of ourselves. When we forget something we believe we should have remembered, we fear the worst, that we are developing dementia. While this is always within the universe of possibilities, and I list a simple dementia test in this endnote1, more often we suffer temporary conditions such as fatigue, anxiety or depression.
If you wish, take a few minutes now for this online memory test. Write your score on a page labeled "memory" in your personal medical records folder. (You have one, don't you? To collect your laboratory work and medical reports should they become important at some point in the future?) You might wish to take the test again when you are tired or anxious, to see the natural variation in your memory. Now you have a baseline, so that you can replace useless worry with actual information. Plus, taking the memory test helps preserve your noggin, as does all mental activity.
Dementia doesn't fall unexpectedly out of the sky. With care, physicians can identify people at risk for dementia. Even ten or twenty years in advance, dementia victims score more poorly on tests of memory and abstract reasoning.2
In a previous newsletter, we outlined the many different forms dementia can take3. Yet each of these develops in the natural environment of neurodegeneration and cognitive loss that takes place over time in all of us. It is this natural degradation that we can work to forestall.
Optimizing Your Genetic Makeup
Section titled “Optimizing Your Genetic Makeup”Microbiologist Bruce Ames PhD has worked in the field of genetics for much of his life. Decades ago he developed the "Ames test" – one of the first tests a pharmaceutical agent has to pass before it is given to humans. The test involves exposing bacteria to the drug, to see if the drug causes mutations in the bacteria.
More recently Dr Ames has turned his attention to humans rather than bacteria and noted that, in many individuals, the aging process results from less than optimal amounts of this or that nutrient. He and his colleagues point out that by giving certain nutrients to human populations, you can, on average, delay aging of that population.
Your biologic car doesn't just fall apart because seventy winters have passed. It falls apart because the mitochondria have been destroyed by free radicals, and because the cell membranes of the brain are short in phosphatidylserine, docosahexaenoic acid (DHA), or any of a number of other substances. It falls apart because decades of microinflammation have destroyed too many neurons or caused too much vascular damage, and because of the toll taken over time by elevated homocysteine or blood sugar, and many similar processes.
We know in detail how these processes occur. In many cases we can increase our supply of the substances we see running low and delay the inevitable.
To those who say you can't improve upon nature, I point to Dr Ames' work. Then I point at the roof, and add that in an entirely natural world we would be wet and cold this February day. I, for one, advocate that we use our knowledge of brain chemistry here presented to preserve our mental functions so that we live as much of our lives as possible with a full bookshelf under that winter cap. This way we can continue to contribute to our world, and chew up fewer resources that might otherwise be devoted to caring for us in our disabled and demented old age. We can try some chemical jujitsu. And then pay nature back with wise living.
Micronutrients and Genetics
Section titled “Micronutrients and Genetics”The major nutrients are the proteins, fats, and carbohydrates. The micronutrients are the vitamins, minerals, and other critical dietary factors.
Our individuality results from the mixing and matching of our genes, our DNA, by which we enjoy some of the qualities of our mothers, and some of the qualities of our fathers. This DNA blueprint results in slightly different versions and slightly different amounts of the proteins that structure us and make us go. We've talked about mitochondria a great deal in this series. Mitochondria vary from person to person. For example, although training and will are critical to a Tour de France bicyclist, only those humans who have a genetic makeup that allows unusually effective mitochondrial function are going to find themselves qualifying for that race.
We reviewed the role of folic acid in brain function, and pointed out that there are great genetic variations in our ability to recycle and re-use folic acid. If a person's genetic make-up includes less-than-optimal versions of enzymes involved in folic acid use, they will be more likely to suffer neurodegeneration and cardiac disease when consuming the amount of folic acid in the normal US diet.
If, however, we give that person an unnaturally high intake of folic acid, this surplus of raw material unloaded on those less-than-optimal enzymes results in a more normal end result. In the case of the folic-acid family of enzymes, we can push homocysteine levels lower than our genetics would normally allow, and thereby reduce damage to blood vessels and to neurons.
The same principle holds with many other organ systems. We may not have been born with the super-effective mitochondria of a Tour de France winner, but if we can arrange to have plenty of the required micronutrients in our system, then the mitochondria we have will function better than they otherwise would.
We've discussed a number of micronutrients in this series. In fact, deprive us completely of any micronutrient and our brain is going to suffer. As an example, we mention iodine nowhere else in this series, but in Kazakhstan, millions of people have IQ's lower by 10 to 15 points due to an unusual dearth of iodine in the diet. Public health workers there are preventing future problems by addition of iodine to people's diets, and I mention this here just to point out that one single magic bullet is not what we lack. What we lack is a comprehensive overview.
Nutrients for Brain Health
Section titled “Nutrients for Brain Health”Multiple Vitamins
Section titled “Multiple Vitamins”A Canadian group4 followed 96 "apparently healthy" senior citizens for one year. At the end of the year they tested the entire group, and compared the results for those who took multiple vitamins and those who did not. The vitamin-supplemented group scored higher on
- short-term memory
- abstract thinking
- working memory
- digit memory
- information processing
- general cognition
There was no difference on any score for the placebo group and no change in the long-term memory scores for either group. I list the ingredients of this vitamin for your interest, and to show you what even a modest vitamin intake can accomplish.
| Vitamin A 400 | retinol equivalents |
| Beta-carotene | 16 milligrams |
| Vitamin B1 | 2.2 milligrams |
| Vitamin B2 | 1.5 milligrams |
| Vitamin B3 | 16 milligrams |
| Vitamin B6 | 3 milligrams |
| Folic acid | 400 micrograms |
| Vitamin B12 | 4 micrograms |
| Vitamin C | 80 milligrams |
| Vitamin D | 4 micrograms |
| Vitamin E | 44 milligrams |
| Iron | 16 milligrams |
| Zinc | 14 milligrams |
| Copper | 1.4 milligrams |
| Selenium | 20 micrograms |
| Iodine | 0.2 milligrams |
| Calcium | 200 milligrams |
| Magnesium | 100 milligrams |
Researchers also measured vitamin levels. Those individuals with lower levels of vitamins in the blood at the beginning of the year and whose levels returned to normal with supplementation improved most. At any point in time, those who had lower vitamin levels scored more poorly than those with adequate levels. None of these people had Alzheimer's.
Antioxidants
Section titled “Antioxidants”Antioxidants are any substance that reduces oxidative damage caused by free radicals.
Because there is much evidence that free radical damage hastens memory loss, mild cognitive impairment, and dementia of several types,5 I recommend action to maintain antioxidant protection. The main line of defense is fresh food, freshly prepared. Fruits and vegetables contain numerous antioxidant substances that we know about and many that we still have yet to identify. In addition I suggest taking the following antioxidant supplements.
Vitamin C
A 1998 study at Rush University in Chicago reported significantly less dementia among a group of 600 older adults taking higher-dose vitamin C and E supplements.6 A study of over 5000 Dutch found diminished rates of Alzheimer's dementia as those who obtained the most vitamin C and E in their diet.7
Among the 15,000 participants in the Nurses' Health Study, considering those aged over 70, those who took vitamins C and E scored best on tests of memory and cognition compared to those nurses who did not take such supplements.8 In a group of 21 patients with Parkinson disease given 3200 units vitamin E and 3000 milligrams vitamin C a day, and followed for at least 6 years, the need to use levodopa (a medication for Parkinson disease that, once started, slowly loses effect) was delayed by an average of 2.5 years.9
Recommended dose: 500 mg of vitamin C a day to protect mitochondria and reduce microinflammation in the blood vessels and brain tissue.
Vitamin E
A study of about 3000 Chicagoans over a three year period showed a 36 percent slower rate of mental decline in the group consuming the most vitamin E, compared to the group consuming the least.10 A similar study showed reduced incidence of Alzheimer's in senior citizens who consumed more vitamin E in their diet.11 Arteriosclerosis contributes mightily to dementia. Vitamin E inhibits the formation of plaque and promotes plaque stability.12 A study in a major medical journal compared vitamin E to selegiline for Alzheimer's disease. Vitamin E slowed the disease better than the pharmaceutical.13
As you are aware from the news media, vitamin E supplements do not always fare well in tests. This is often because researchers use alpha-tocopherol, instead of mixed tocopherols. Mixed tocopherols are more effective than alpha-tocopherol alone.14
Recommended dose: 400 mg daily to protect mitochondria and reduce microinflammation in the blood vessels and brain tissue. Mixed tocopherols are best, in the nuts, green leafy vegetables, and vegetable oils you eat every day. Right?
Lipoic acid
Lipoic acid helps recycle all other antioxidants. One scientist concluded that lipoic acid provides "the most remarkable effect of a therapeutic substance in protection against cerebral ischemia-reperfusion so far known.15" "Ischemia-reperfusion" is medical shorthand for the damage to the brain due to stroke.
Animal studies showed a 225 percent increase in free radicals following stroke, which is reduced to just 60 percent when the animals have been taking lipoic acid each day.
Recommended dose: 100 mg a day.
B-Vitamins
Section titled “B-Vitamins”Vitamin B12
Section titled “Vitamin B12”We know that vitamin B12 deficiency impairs intellect. Homocysteine rises when B12 is low, and high homocysteine occurs in concert with dementia.16 While there is no rock-solid evidence that lowering homocysteine prevents dementia, vitamin B12 deficiency is common as we age and can be devastating to the gray matter. In our earlier discussion of vitamin B12, we noted that as B12 activity decreases, a substance called methylmalonic acid can accumulate even though your doctor's laboratory reports "normal' B12 levels. When we reach the age of 70 to 90, methylmalonic acid is high in about 40 percent of us.17 The higher it is, the more poorly we score on tests of memory and judgment.18
B12 deficiency is more common in elderly white men, least common in black and Asian-American women.19 Even in "high-functioning" elderly Chinese, high levels of homocysteine were associated with cognitive deficits.20
Recommended dose: It seems to me prudent to take vitamins B12 and folic acid while the researchers gather their data.21 I suggest about 1000 mcg daily of cyano-cobalamin, or 400 mcg of the more active adenosyl- or methyl-cobalamin. (All these are forms of vitamin B12) For those with existing memory or cognitive problems, I suggest a therapeutic trial of vitamin B12 injections. Taking B12 without folic acid can cause neurologic injury; don't do it.
Folic acid
Section titled “Folic acid”The evidence: Folic acid protects not just from dementia, but from other manifestations of vascular disease and cancer. Low folic acid correlates with impaired cognition.22 While spinach and yeast are the foods richest in folic acid, we absorb only about half the folic acid in these foods. Studies show we are much more likely to benefit from folic acid in supplement form.23
Recommended dose: 400 to 800 micrograms per day. You will find this in most multiple vitamins. Taking folic acid when you are B12 deficient can promote dementia. Commonly, B12 deficiency occurs because we don't absorb the vitamin. Periodic check-ups and lab work should identify this.
Niacin
Section titled “Niacin”Niacin deficiency, called pellagra, has long been known for its "three D's"- dermatitis, dementia, diarrhea. Could long-term subclinical niacin deficiency lead to dementia? Suffice to say that people who consume more niacin have less dementia.
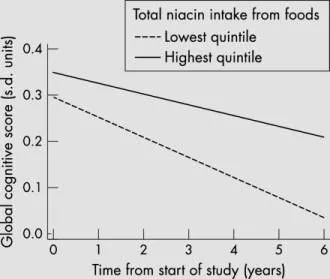
Figure 1 24
Researchers followed about 3000 senior citizens for six years. Those in the highest fifth of niacin intake had slower rates of cognitive decline than those in the lowest fifth. This graph excludes people with known vascular disease, which has its own effect on mental decline and dilutes the difference due to niacin.25 The "high niacin" group obtained over 36 milligrams niacin per day from both food and supplements.
Get Your Vitamins
Section titled “Get Your Vitamins”A multiple vitamin/mineral supplement can provide the nutrients listed above. We may not need everything in that multiple vitamin, but with no cost-effective way to know, that extra zinc, magnesium, and other B vitamins may come in handy. So, which multiple/vitamin should you take? First, as shown above, even a simple daily vitamin provides some benefit.
Other Micronutrients
Section titled “Other Micronutrients”Our mitochondria are doing a great deal of chemistry to make us go, involving many different reactions which require biotin, copper, pantothenic acid, and vitamin B6.26
Iron
Although it is well known that iron excess can lead to oxidant damage, it is also true that iron deficiency impairs mitochondrial function and leads to neurodegeneration.27 Take iron only if you are deficient, as excessive iron is harmful.
Zinc
Zinc deficiency disrupts mitochondrial function. About 10 percent of the US population consumes less than 50 percent of the RDA for zinc.28 You should obtain zinc in your daily vitamin/mineral.
Cognitive Enhancers
Section titled “Cognitive Enhancers”Acetyl-l-carnitine increases synthesis of membrane phospholipids, as well as increasing activity of nerve growth factors.29
Recommended dose: While many studies used a higher dose, I suggest 500 mg daily as a reasonable compromise between efficacy and cost.
Phosphatidlyserine
Phosphatidylserine is critical to the function of neurons.
Recommended dose: While many studies used a higher dose, I suggest 100 mg daily as a reasonable compromise between efficacy and cost.
Ginkgo biloba
The leaves of the Ginkgo tree have been used for medical purposes for millennia. Chinese medical texts dating from before the advent of statistics describe the ginkgo leaf as a "benefit to the brain". Ginkgo dilates blood vessels, has antioxidant properties, and reduces the tendency to form blood clots. In addition, it reduces anxiety in healthy young adults as well as elderly people with cognitive decline.
Of the many studies of ginkgo for Alzheimer's dementia, most are small studies showing a modest benefit. When studying people with severe dementia, often the ginkgo group will have experienced no change in function, while the placebo group has deteriorated. In people with mild to moderate dementia, the Italians recently compared ginkgo to Aricept (the most commonly used pharmaceutical agent for dementia). Both ginkgo and the drug produced equivalent but modest benefit, and either was better than placebo.30
The great majority of the published studies have shown benefit for people with age-associated memory loss. An effective form of ginkgo (special extract EGb 761) alone can delay the onset of Alzheimer's dementia by about 2 years.31
Because ginkgo slows the formation of blood clots, some physicians recommend against taking it along with anticoagulants, although recent studies show no interaction between ginkgo and coumadin32 or aspirin.
Recommended dose: While many studies used doses over 120 mg, I suggest 40 to 60 mg daily as a reasonable compromise between efficacy and cost.
Dietary Fat
Section titled “Dietary Fat”Fish or Fish Oil
Section titled “Fish or Fish Oil”Fish oil reduces microinflammation and normalizes function of neural membranes.
Omega-3 fatty acids
Section titled “Omega-3 fatty acids”Previous newsletters outlined major mechanisms of dementia. As one reviewer noted, the fats we eat can affect brain function through "atherosclerosis and thrombosis, inflammation, via an effect on brain development and membrane functioning, or via accumulation of beta-amyloid".33 That makes a lot of ways one single nutrient class can help. The largest study of dietary fats involved over 5000 people in Rotterdam in the Netherlands. Those who consumed the most omega-3 fatty acids, as in fish, had only 40 percent the rate of dementia as those who consumed the least. Conversely, the "bad fats" such as trans fats and saturated fats increased the risk of dementia.34 I hate to tell you that the same group published a later report that these associations seemed to disappear with time.35 However, researchers are approaching their sixtieth year of observation of over 5000 people in Framingham, Massachusetts. This study, much longer than the still-equivocal Rotterdam study, found that people who ate about three servings of fish a week had about half the incidence of dementia as those who did not.36
A study of 2,500 Chicagoans also showed benefit37, as have most of the smaller studies.38 Indeed, even looking at mild cognitive decline short of dementia, fish oils help.39 When researchers looked at 1600 younger people aged 45 to 70, higher fish oil consumption was still highly correlated with improved cognitive function.40 So, most researchers in this field believe that omega-3 fatty acids reduce the risk of cognitive decline and Alzheimer's.
Omega-3 fatty acids are incorporated into critical cell membrane constituents such as phophatidylcholine and phosphatidylserine. While we all know in an abstract way that what we eat forms what we are, the brain has been shown very clearly to contain more omega-3 fatty acid when we consume more omega-3 fatty acid in our diet.41 Research finds that when levels of the most important omega-3 fatty acids, EPA and DHA, are low in the blood, that people are more likely to develop Alzheimer's. In fact, older people with cognitive impairment, but no dementia, also have low levels of EPA and DHA.42
In summary, omega-3 fatty acids
- reduce microinflammation;
- reduce tendency of blood to clot and impair blood flow to the brain;
- improve function of joints, which will allow you to exercise which protects your brain;
- improve brain membrane function;
- reduce tendency to form beta-amyloid.
Recommended dose: For men, I suggest two teaspoons of cod liver oil a day, which should provide about 800 to 1000 milligrams each of EPA and DHA. Women might wind up with excessive vitamin A which is in cod liver oil, (which might hasten bone loss), and might do better with omega-3 in capsule form. (Cod liver oil contains vitamins A and D as well as the omega-3 oils).
Trans fats
Section titled “Trans fats”Trans fats have opposite effects, as noted above.43 Avoid ‘em. Recognize that the FDA allowsproducers to label their food as "zero trans fat" if the food contains less than 500 milligrams of trans fats per serving. You eat five servings of such foods, and you might have consumed up to about 20 calories of trans fat. Caveat emptor.
Hazards to Avoid
Section titled “Hazards to Avoid”First, remember that certain conditions can accelerate cognitive decline.
- Gluten intolerance
- Magnesium deficiency 44
- Alcohol overuse
- Concentrated carbohydrates
- Trauma to the head. (Don't take up boxing. Wear a helmet when appropriate.)
You cannot avoid a family history of dementia or Parkinson's disease: that die stopped spinning long ago. But if you do have such relatives, remember that you can give your brain the raw materials it needs to keep itself in shape. Feed it nutrients and feed it physical and mental exercise.
Coming Next Month
Section titled “ Coming Next Month”Next month I will conclude this series with
- recommendations and evidence for other (and very important) measures that prevent brain degeneration;
- "second-line" nutrients;
- prescription drugs that slow brain degeneration as a side-effect;
- an economic analysis;
- reliable brands of supplements mentioned above.
Satisfaction Guaranteed Referral Policy
Section titled “Satisfaction Guaranteed Referral Policy”In this office, are careful about where we refer patients, because if they are not happy with the specialist we send them to, that reflects poorly on us. We'd like to make it easy for you to refer your friends and family to us. To that end, if you refer a friend or family member to our office for a first-time visit, we'll give them a "satisfaction-guaranteed visit." That means if we do not meet or exceed their expectations we will refund their office fee.
We make this offer only for people who are not yet patients here and only for those referred by current patients.
For your information and for referral purposes, here is our general philosophy in four parts:
1. We try hard to give you a plan that makes you, as much as possible, independent of the healthcare system. For example, non-pharmaceutical remedies don't require physician visits for monitoring and refills. With pharmaceuticals, we make your prescriptions maximally refillable in order to reduce the frequency of office visits.
2. We work to find a fundamental cause for illness, so that people can get well if possible and get off the medical care merry-go-round.
3. We help you enhance the strengths and good health habits you already have. Lifestyle changes that reduce blood pressure, blood sugar, and cholesterol involve no further medical costs.
4. We do not bill your insurance for you, but we can give you the requisite form including diagnosis, treatment, and fee, which you can send to your insurance for a reimbursement.
Thank you for your interest in our newsletter and our patient-based medical practice.
Endnotes:
Section titled “Endnotes:”1 The most common test a primary care physician or general neurologist will use for dementia is the Mini Mental State Examination. A simpler test is the Mini-Cog. Developed by Soo Borson M.D. and her colleagues at the University of Washington, and reported in the International Journal Of Geriatric Psychiatry 2000 15:21, the test combines the clock drawing test (CDT) with a three item recall test.
To perform the clock drawing test, the patient is given a circle and is asked to draw a clock and then put the hands on the clock to make it read 2.45. To quote Dr Borson, "We considered the CDT normal if all numbers were present in the correct sequence and position and the hands readably displayed the requested time. The Mini-Cog was constructed by combining the CDT with uncued three-word recall derived from the CASI. To assess testing time, 44 elderly subjects, (26 with a variety of dementing disorders, 18 with depression or anxiety or who were caregivers) were asked to repeat three unrelated words (possible score 0-3) then to draw a clock (scored as normal or abnormal) which served simultaneously as the recall distracter, and then to recall the three previously presented items." If no items were recalled, the test indicated dementia, if three items were recalled, the diagnosis was "no dementia." If one or two items were recalled, the CDT was scored and the decision was based on that result.
Recall 0 items – dementia
Recall 1 or 2, do CDT and go by that
Recall 3, non-demented
2 The preclinical phase of alzheimer disease: A 22-year prospective study of the Framingham Cohort. Arch Neurol 2000 Jun;57(6):808-13
Department of Mathematics and Statistics, Statistical Consulting Unit, 111 Cummington St, Boston University College of Arts and Sciences, Boston, MA 02215, USA. mfelias@aol.com
OBJECTIVES: To relate performance on tests of cognitive ability to the subsequent development of probable Alzheimer disease (pAD) and to identify the pattern of earliest changes in cognitive functioning associated with a diagnosis of pAD. DESIGN: From May 1975 to November 1979, a screening neuropsychological battery was administered to Framingham Study participants. They were followed up prospectively for 22 years and examined at least every 2 years for the development of pAD. SETTING: A community-based center for epidemiological research. PARTICIPANTS: Subjects were 1076 participants of the Framingham Study aged 65 to 94 years who were free of dementia and stroke at baseline (initial) neuropsychological testing. MAIN OUTCOME MEASURE: Presence or absence of pAD during a 22-year surveillance period was related to test performance at initial neuropsychological testing. RESULTS: Lower scores for measures of new learning, recall, retention, and abstract reasoning obtained during a dementia-free period were associated with the development of pAD. Lower scores for measures of abstract reasoning and retention predicted pAD after a dementia-free period of 10 years. CONCLUSIONS: The "preclinical phase" of detectable lowering of cognitive functioning precedes the appearance of pAD by many years. Measures of retention of information and abstract reasoning are among the strongest predictors of pAD when the interval between initial assessment and the development of pAD is long.
3 And I have purposely omitted discussion of certain genetic factors the influence our risk of dementia, such as Apo E. The measures I will recommend should delay the onset of dementia in those with a genetic predisposition. At this time, though we can tell you if you have certain genetic markers for dementia, we don't do anything differently as a result.
4 "Effect of Vitamin and Trace-Element Supplementation on Cognitive Function in Elderly Subjects" Chandra RK Nutrition 2001 17:709
5 There are a raft of studies on antioxidants and prevention of dementia. Everyone who studies the brain knows how oxidative damage can easily occur in that rapidly metabolizing tissue, and how prone the delicate threads of the brain are. Results are confusing because everyone designs their study differently, uses different antioxidants, different doses of antioxidants, different forms of those antioxidants, and may study antioxidants in foods, in supplements or both combined. Bottom line is, antioxidants do help prevent dementia to a degree. Here is just one of thousands of studies.
Lipid peroxidation is an early event in the brain in amnestic mild cognitive impairment. Ann Neurol 2005 Nov;58(5):730-5
Multiple studies demonstrate that the brain in Alzheimer's disease (AD) contains extensive oxidative damage. Most of these studies used advanced-stage AD patients raising the question of whether oxidative damage is a late effect of neurodegeneration or precedes and contributes to the pathogenesis of AD. Here we describe F(2)-isoprostane (F(2)-IsoP) and F(4)-neuroprostane (F(4)-NP) levels in longitudinally followed, well documented autopsied normal control subjects and patients with amnestic mild cognitive impairment (MCI), and late-stage AD. Gas chromatography/negative ion chemical ionization/mass spectrometry was used to determine F(2)-IsoP and F(4)-NP levels. Significant increases in F(2)-IsoP levels were found in frontal, parietal and occipital lobes in MCI and late AD compared to controls but no significant differences were present between MCI and late AD. A significant increase in F(4)-NPs was present in parietal and occipital lobes in MCI compared to controls and a significant increase was present in these regions and hippocampus in late AD compared to controls. The only difference between MCI and late AD was significantly increased F(4)-NP in hippocampus in late AD. Our data indicate that lipid peroxidation is present in the brain of MCI patients and suggest that oxidative damage may play a role in the pathogenesis of AD.
6 Vitamin E and vitamin C supplement use and risk of incident Alzheimer disease. Alzheimer Dis Assoc Disord 1998 Sep;12(3):121-6
Rush Institute for Healthy Aging and Rush Alzheimer's Disease Center, Rush University, Chicago, Illinois, USA.
Oxidative stress may play a role in neurologic disease. The present study examined the relation between use of vitamin E and vitamin C and incident Alzheimer disease in a prospective study of 633 persons 65 years and older. A stratified random sample was selected from a disease-free population. At baseline, all vitamin supplements taken in the previous 2 weeks were identified by direct inspection. After an average follow-up period of 4.3 years, 91 of the sample participants with vitamin information met accepted criteria for the clinical diagnosis of Alzheimer disease. None of the 27 vitamin E supplement users had Alzheimer disease compared with 3.9 predicted based on the crude observed incidence among nonusers (p = 0.04) and 2.5 predicted based on age, sex, years of education, and length of follow-up interval (p = 0.23). None of the 23 vitamin C supplement users had Alzheimer disease compared with 3.3 predicted based on the crude observed incidence among nonusers (p = 0.10) and 3.2 predicted adjusted for age, sex, education, and follow-up interval (p = 0.04). There was no relation between Alzheimer disease and use of multivitamins. These data suggest that use of the higher-dose vitamin E and vitamin C supplements may lower the risk of Alzheimer disease.
7 Engelhart MJ,Geerlings MI,Ruitenberg A,van Swieten JC,Hofman A,Witteman JC,Breteler MM. "Dietary intake of antioxidants and risk of Alzheimer disease." Journal of the American Medical Association. 2002 Jun 26;287:3223-9. (Issue number 24) (Copy available). Copy in office. 102112509 Research reported by Department of Epidemiology and Biostatistics, Erasmus Medical Center, 3000 DR Rotterdam, The Netherlands.. =18974= = Conclusion: High dietary intake of vitamin C and vitamin E may lower the risk of Alzheimer disease. = Author's abstract: CONTEXT: Laboratory findings have suggested that oxidative stress may contribute to the pathogenesis of Alzheimer disease. Therefore, the risk of Alzheimer disease might be reduced by intake of antioxidants that counteract the detrimental effects of oxidative stress. OBJECTIVE: To determine whether dietary intake of antioxidants is related to risk of Alzheimer disease. DESIGN AND SETTING: The Rotterdam Study, a population-based, prospective cohort study conducted in the Netherlands. PARTICIPANTS: A total of 5395 participants who, at baseline (1990-1993), were aged at least 55 years, free of dementia, and noninstitutionalized and had reliable dietary assessment. Participants were reexamined in 1993-1994 and 1997-1999 and were continuously monitored for incident dementia. MAIN OUTCOME MEASURES: Incidence of Alzheimer disease, based on Diagnostic and Statistical Manual of Mental Disorders, Revised Third Edition (DSM-III-R) criteria and National Institute of Neurological and Communicative Disorders and Stroke and Alzheimer Disease and Related Disorders Association (NINCDS-ADRDA) criteria, associated with dietary intake of beta carotene, flavonoids, vitamin C, and vitamin E. RESULTS: After a mean follow-up of 6 years, 197 participants developed dementia, of whom 146 had Alzheimer disease. When adjustments were made for age, sex, baseline Mini-Mental State Examination score, alcohol intake, education, smoking habits, pack-years of smoking, body mass index, total energy intake, presence of carotid plaques, and use of antioxidative supplements, high intake of vitamin C and vitamin E was associated with lower risk of Alzheimer disease (rate ratios [RRs] per 1-SD increase in intake were 0.82 [95% confidence interval [CI], 0.68-0.99] and 0.82 [95% CI, 0.66-1.00], respectively). Among current smokers, this relationship was most pronounced (RRs, 0.65 [95% CI, 0.37-1.14] and 0.58 [95% CI, 0.30-1.12], respectively) and also was present for intake of beta carotene (RR, 0.49 [95% CI, 0.27-0.92]) and flavonoids (RR, 0.54 [95% CI, 0.31-0.96]). The associations did not vary by education or apolipoprotein E genotype.
8 Grodstein F,Chen J,Willett WC. "High-dose antioxidant supplements and cognitive function in community-dwelling elderly women." American Journal of Clinical Nutrition. 2003 Apr;77:975-84. (Issue number 4) Research reported by Channing Laboratory, Department of Medicine, Brigham and Women's Hospital, Harvard Medical School, Boston, MA 02115, USA. = Conclusion: The use of specific vitamin E supplements, but not specific vitamin C supplements, may be related to modest cognitive benefits in older women. = Author's abstract: BACKGROUND: Experimental data suggest that oxygen free radicals are probably involved in the deterioration of cognitive processes. OBJECTIVE: Our objective was to investigate the relation of high-dose antioxidant supplements to cognition. DESIGN: Information on the use of specific supplements containing vitamins E and C was collected biennially via mailed questionnaires beginning in 1980 from 14 968 community-dwelling women who participated in the Nurses' Health Study. From 1995 to 2000, telephone tests of cognitive function [Telephone Interview of Cognitive Status (TICS), delayed recall of the TICS 10-word list, immediate and delayed recall of a short paragraph, a test of verbal fluency, and a digit span backwards test] were administered to the women, who were 70-79 y of age at that time. We used linear and logistic regression models to calculate multivariate-adjusted mean differences in test scores and relative risks of a low score for specific supplement users compared with nonusers. RESULTS: Long-term, current users of vitamin E with vitamin C had significantly better mean performance, as judged by a global score that combined individual test scores, than did women who had never used vitamin E or C (P = 0.03); there was a trend for increasingly higher mean scores with increasing durations of use (P = 0.04). These associations were strongest among women with low dietary intakes of alpha-tocopherol. Benefits were less consistent for women taking vitamin E alone, with no evidence of higher scores with longer durations of use. Use of specific vitamin C supplements alone had little relation to performance on our cognitive tests.
9 High doses of vitamin E in the treatment of disorders of the central nervous system in the aged Govind T Vatassery et al Am J Clin Nutr 1999;70:793–801.
10 Vitamin E and cognitive decline in older persons. Arch Neurol 2002 Jul;59(7):1125-32
Department of Preventive medicine, Rush Institute for Healthy Aging, Rush-Presbyterian-St Luke's Medical Center, 1645 W Jackson, Suite 675, Chicago, IL 60612, USA.
BACKGROUND: Previous studies raise the possibility that antioxidants protect against neurodegenerative diseases. OBJECTIVE: To examine whether intake of antioxidant nutrients, including vitamin E, vitamin C, and carotene, is associated with reduced cognitive decline with age. DESIGN: Longitudinal population-based study conducted from September 17, 1993, to November 20, 2000, with an average follow-up of 3.2 years. PATIENTS: The patients were 2889 community residents, aged 65 to 102 years, who completed a food frequency questionnaire, on average 18 months after baseline. MAIN OUTCOME MEASURE: Cognitive change as measured by 4 tests (the East Boston Memory Test, which tests immediate and delayed recall; the Mini-Mental State Examination; and the Symbol Digit Modalities Test) at baseline and 3 years for all participants, and at 6 months for 288 randomly selected participants. RESULTS: We used random-effects models to estimate nutrient effects on individual change in the average score of the 4 cognitive tests. The cognitive score declined on average by 5.0 x 10(-2) standardized units per year. There was a 36% reduction in the rate of decline among persons in the highest quintile of total vitamin E intake (-4.3 x 10(-2) standardized units per year) compared with those in the lowest quintile (-6.7 x 10(-2) standardized units per year) (P =.05), in a model adjusted for age, race, sex, educational level, current smoking, alcohol consumption, total calorie (energy) intake, and total intakes of vitamin C, carotene, and vitamin A. We also observed a reduced decline with higher vitamin E intake from foods (P =.03 for trend). There was little evidence of association with vitamin C or carotene intake. CONCLUSION: Vitamin E intake, from foods or supplements, is associated with less cognitive decline with age.
11 Morris MC,Evans DA,Bienias JL,Tangney CC,Bennett DA,Aggarwal N,Wilson RS,Scherr PA,others. "Dietary intake of antioxidant nutrients and the risk of incident Alzheimer disease in a biracial community study." Journal of the American Medical Association. 2002 Jun 26;287:3230-7. (Issue number 24) Research reported by Rush Institute for Healthy Aging, Rush-Presbyterian-St Luke's Medical Center, 1645 W Jackson, Suite 675, Chicago, IL 60612, USA. mmorris@rush.edu. =18976= = Conclusion: This study suggests that vitamin E from food, but not other antioxidants, may be associated with a reduced risk of AD. Unexpectedly, this association was observed only among individuals without the APOE epsilon 4 allele. = Author's abstract: CONTEXT: Oxidative processes have been suggested as elements in the development of Alzheimer disease (AD), but whether dietary intake of vitamin E and other antioxidant nutrients prevents its development is unknown. OBJECTIVE: To examine whether intake of antioxidant nutrients, vitamin E, vitamin C, and beta carotene is associated with incident AD. DESIGN, SETTING, AND PARTICIPANTS: Prospective study, conducted from 1993 to 2000, of individuals selected in a stratified random sample of community-dwelling residents. The 815 residents 65 years and older were free of AD at baseline and were followed up for a mean of 3.9 years. They completed food frequency questionnaires an average of 1.7 years after baseline. MAIN OUTCOME MEASURE: Incident AD diagnosed in clinical evaluations with standardized criteria. RESULTS: Increasing vitamin E intake from foods was associated with decreased risk of developing AD after adjustment for age, education, sex, race, APOE epsilon 4, and length of follow-up. Relative risks (95% confidence intervals [CIs]) from lowest to highest quintiles of intake were 1.00, 0.71 (0.24-2.07), 0.62 (0.26-1.45), 0.71 (0.27-1.88), and 0.30 (0.10-0.92) (P for trend =.05). The protective association of vitamin E was observed only among persons who were APOE epsilon 4 negative. Adjustment for other dietary factors reduced the protective association. After adjustment for baseline memory score, the risk was 0.36 (95% CI, 0.11-1.17). Intake of vitamin C, beta carotene, and vitamin E from supplements was not significantly associated with risk of AD.
12 High doses of vitamin E in the treatment of disorders of the central nervous system in the aged Govind T Vatassery et al Am J Clin Nutr 1999;70:793–801. "platelet adhesion was inhibited by vitamin E in dosages of 400 IU/d. Similarly, vitamin E inhibited the adhesion of leukocytes to endothelium and promoted plaque stability, as reviewed by Diaz et al (49). In addition, a-tocopherol inhibited smooth muscle cell proliferation (50). In this case, a-tocopherol inhibited protein kinase C and smooth muscle cell proliferation along with activating protein phosphatase in a dose-dependent manner." Page 797
13 "A Controlled Trial Of Selegiline, Alpha-Tocopherol, Or Both As Treatment For Alzheimer's Disease" Sano M. N Engl J Med 1997;336:1216-22.
14 Relation of the tocopherol forms to incident Alzheimer disease and to cognitive change. Am J Clin Nutr 2005 Feb;81(2):508-14
Rush Institute for Healthy Aging, Rush University Medical Center, Chicago, IL 60612, USA.
BACKGROUND: High intake of vitamin E from food (tocopherol), but not from supplements (which usually contain alpha-tocopherol), is inversely associated with Alzheimer disease. OBJECTIVE: We examined whether food intakes of vitamin E, alpha-tocopherol equivalents (a measure of the relative biologic activity of tocopherols and tocotrienols), or individual tocopherols would protect against incident Alzheimer disease and cognitive decline over 6 y in participants of the Chicago Health and Aging Project. DESIGN: The 1993-2002 study of community residents aged >or=65 y included the administration of 4 cognitive tests and clinical evaluations for Alzheimer disease. Dietary assessment was by food-frequency questionnaire. RESULTS: Tocopherol intake from food was related to the 4-y incidence of Alzheimer disease determined by logistic regression in 1041 participants who were clinically evaluated (n=162 incident cases) and to change in a global cognitive score determined by mixed models in 3718 participants. Higher intakes of vitamin E (relative risk: 0.74 per 5 mg/d increase; 95% CI: 0.62, 0.88) and alpha-tocopherol equivalents (relative risk: 0.56 per 5 mg/d increase; 95% CI: 0.32, 0.98) were associated with a reduced incidence of Alzheimer disease in separate multiple-adjusted models that included intakes of saturated and trans fats and docosahexaenoic acid. alpha- and gamma-Tocopherol had independent associations. In separate mixed models, a slower rate of cognitive decline was associated with intakes of vitamin E, alpha-tocopherol equivalents, and alpha- and gamma-tocopherols. CONCLUSION: The results suggest that various tocopherol forms rather than alpha- tocopherol alone may be important in the vitamin E protective association with Alzheimer disease.
15 Free Radical Biology & Medicine 1997 22;1:359
16 Plasma homocysteine as a risk factor for dementia and Alzheimer's disease. N Engl J Med 2002 Feb 14;346(7):476-83
Department of Neurology, Boston University School of Medicine, MA 02118-2526, USA.
BACKGROUND: In cross-sectional studies, elevated plasma homocysteine levels have been associated with poor cognition and dementia. Studies of newly diagnosed dementia are required in order to establish whether the elevated homocysteine levels precede the onset of dementia or result from dementia-related nutritional and vitamin deficiencies. METHODS: A total of 1092 subjects without dementia (667 women and 425 men; mean age, 76 years) from the Framingham Study constituted our study sample. We examined the relation of the plasma total homocysteine level measured at base line and that measured eight years earlier to the risk of newly diagnosed dementia on follow-up. We used multivariable proportional-hazards regression to adjust for age, sex, apolipoprotein E genotype, vascular risk factors other than homocysteine, and plasma levels of folate and vitamins B12 and B6. RESULTS: Over a median follow-up period of eight years, dementia developed in 111 subjects, including 83 given a diagnosis of Alzheimer's disease. The multivariable-adjusted relative risk of dementia was 1.4 (95 percent confidence interval, 1.1 to 1.9) for each increase of 1 SD in the log-transformed homocysteine value either at base line or eight years earlier. The relative risk of Alzheimer's disease was 1.8 (95 percent confidence interval, 1.3 to 2.5) per increase of 1 SD at base line and 1.6 (95 percent confidence interval, 1.2 to 2.1) per increase of 1 SD eight years before base line. With a plasma homocysteine level greater than 14 micromol per liter, the risk of Alzheimer's disease nearly doubled. CONCLUSIONS: An increased plasma homocysteine level is a strong, independent risk factor for the development of dementia and Alzheimer's disease.
17 Am J Clin Nutr 2006;84:1406
18 Assessing the association between vitamin B-12 status and cognitive function in older adults. Joshua W Miller Am J Clin Nutr 2006;84:1259–60.
19 Serum cobalamin, homocysteine, and methylmalonic acid concentrations in a multiethnic elderly population: ethnic and sex differences in cobalamin and metabolite abnormalities Ralph Carmel, et al Am J Clin Nutr 1999; 70:904–10.
20 Homocysteine, folate, and vitamin B-12 and cognitive performance in older Chinese adults: findings from the Singapore Longitudinal Ageing Study. Lei Feng et al Am J Clin Nutr 2006;84:1506 –12.
21 Prevalence of cobalamin deficiency in the Framingham elderly population. Am J Clin Nutr 1994 Jul;60(1):2-11
Department of Medicine, Columbia-Presbyterian Medical Center, New York, NY 10032.
Abstract: To determine whether the increased prevalence of low serum cobalamin concentrations in elderly people represents true deficiency, serum concentrations of cobalamin and folate and of metabolites that are sensitive indicators of cobalamin deficiency were measured in 548 surviving members of the original Framingham Study cohort. Serum cobalamin concentrations < 258 pmol/L were found in 222 subjects (40.5%) compared with 17.9% of younger control subjects (P < 0.001). Serum methylmalonic acid and total homocysteine concentrations were markedly elevated in association with cobalamin values < 258 pmol/L in 11.3% and 5.7%, respectively, of the cohort. Both metabolites were increased in 3.8% of the cohort, associated with significantly lower erythrocyte counts and higher mean cell volumes. Serum metabolites correlated best with serum cobalamin values, even when subnormal determinations were excluded. The prevalence of cobalamin deficiency was > or = 12% in a large sample of free-living elderly Americans. Many elderly people with "normal" serum vitamin concentrations are metabolically deficient in cobalamin or folate.
22 Am J Clin Nutr 2006;84:1406
See also "Low folate status is associated with impaired cognitive function and dementia in the Sacramento Area Latino Study on Aging"
Marisa I Ramos et al * * Am J Clin Nutr 2005;82:1346–52.
23 "Determining bioavailability of food folate" Mary P Hannon-Fletcher et al Am J Clin Nutr 2004;80:911- 8.
ABSTRACT Background: The concept of dietary folate equivalents (DFEs) in the United States recognizes the differences in bioavailability between natural food folates and the synthetic vitamin, folic acid. However, many published reports on folate bioavailability are problematic because of several confounding factors. Objective:Wecompared the bioavailability of food folates with that of folic acid under controlled conditions. To broadly represent the extent to which natural folates are conjugated in foods, we used 2 natural sources of folate, spinach (50% polyglutamyl folate) and yeast (100% polyglutamyl folate). Design: Ninety-six men were randomly assigned according to their screening plasma homocysteine (tHcy) concentration to 1 of 4 treatment groups for an intervention period of 30 d. Each subject received (daily under supervision) either a folate-depleted "carrier" meal or a drink plus 1) placebo tablet, 2) 200 _g folic acid in a tablet, 3) 200 _g natural folate provided as spinach, or 4) 200 _g natural folate provided as yeast. Results: Among the subjects who completed the intervention, responses (increase in serum folate, lowering of tHcy) relative to those in the placebo group (n_18) were significant in the folic acid group (n _ 18) but not in the yeast folate (n _ 19) or the spinach folate (n _ 18) groups. Both natural sources of folate were significantly less bioavailable than was folic acid. Overall estimations of folate bioavailability relative to that of folic acid were found to be between 30% (spinach) and 59% (yeast). Conclusion: Relative bioavailability estimates were consistent with the estimates from the metabolic study that were used as a basis to derive the US DFE value.
24 Change in cognitive score (z score) for participants in quintiles 1 and 5 of niacin intake from foods based on the basic adjusted model (table 3) among 2824 participants with no history of stroke or myocardial infarction. The figure data were computed for a 75 year old female with 12 years of education. The annual rate of change in cognitive score was –0.048 SU/year in the lowest quintile of niacin intake (median = 12.6 milligrams/day) and –0.027 SU/year in the highest quintile of intake (median = 22.1 milligrams/day). The difference in rates of 0.021 SU/year was statistically significant at p = 0.003. The rate differences for quintiles 2–4 were 0.003, 0.002, and 0.009, respectively, and were not statistically significant." Source: http://jnnp.bmjjournals.com/cgi/content/full/75/8/1093
25 Dietary niacin and the risk of incident Alzheimer's disease and of cognitive decline. J Neurol Neurosurg Psychiatry 2004 Aug;75(8):1093-9
Rush Institute for Healthy Aging, Centers for Disease Control and Prevention, Atlanta, GA, USA.
BACKGROUND: Dementia can be caused by severe niacin insufficiency, but it is unknown whether variation in intake of niacin in the usual diet is linked to neurodegenerative decline. We examined whether dietary intake of niacin was associated with incident Alzheimer's disease (AD) and cognitive decline in a large, prospective study. METHODS: This study was conducted in 1993-2002 in a geographically defined Chicago community of 6158 residents aged 65 years and older. Nutrient intake was determined by food frequency questionnaire. Four cognitive tests were administered to all study participants at 3 year intervals in a 6 year follow up. A total of 3718 participants had dietary data and at least two cognitive assessments for analyses of cognitive change over a median 5.5 years. Clinical evaluations were performed on a stratified random sample of 815 participants initially unaffected by AD, and 131 participants were diagnosed with 4 year incident AD by standardised criteria. RESULTS: Energy adjusted niacin intake had a protective effect on development of AD and cognitive decline. In a logistic regression model, relative risks (95% confidence intervals) for incident AD from lowest to highest quintiles of total niacin intake were: 1.0 (referent) 0.3 (0.1 to 0.6), 0.3 (0.1 to 0.7), 0.6 (0.3 to 1.3), and 0.3 (0.1 to 0.7) adjusted for age, sex, race, education, and ApoE e4 status. Niacin intake from foods was also inversely associated with AD (p for linear trend = 0.002 in the adjusted model). In an adjusted random effects model, higher food intake of niacin was associated with a slower annual rate of cognitive decline, by 0.019 standardised units (SU) per natural log increase in intake (mg) (p = 0.05). Stronger associations were observed in analyses that excluded participants with a history of cardiovascular disease (beta = 0.028 SU/year; p = 0.008), those with low baseline cognitive scores (beta = 0.023 SU/year; p = 0.02), or those with fewer than 12 years' education (beta = 0.035 SU/year; p = 0.002) CONCLUSION: Dietary niacin may protect against AD and age related cognitive decline.
26 Delaying the Mitochondrial Decay of Aging. Ann N Y Acad Sci 2004 1019:406
27 Delaying the Mitochondrial Decay of Aging. Ann N Y Acad Sci 2004 1019:406
28 Delaying the Mitochondrial Decay of Aging. Ann N Y Acad Sci 2004 1019:406
29 Effects of nerve growth factor and acetyl-L-carnitine arginyl amide on the human neuronal line HCN-1A. Int J Dev Neurosci 1992 Oct;10(5):361-73
The HCN-1A clonal cell line, derived from the cortical tissue of a patient with unilateral megencephaly, was shown to differentiate into a mature neuronal-like state in the presence of the nerve growth factor, dibutyryl cyclic adenosine, 3',5'-monophosphate and either 1-isobutyl-3-methylxanthine or forskolin. Differentiation was assessed by measuring the percentage of cells that displayed branched, varicose processes that stained for synaptophysin. Treatment of cultures with a cocktail containing forskolin increased immunocytochemical staining for gamma aminobutyric (GABA), neurofilament protein and the nerve growth factor receptor species p75NGFR. Treatment with acetyl-L-carnitine alone had some effects on the cell morphology while acetyl-L-carnitine arginyl amide and nerve growth factor together increased the GABA content. Positive staining levels for the neurotransmitters gamma aminobutyric acid, glutamate, somatostatin, cholecystokinin and vasoactive intestinal polypeptide were measured quantitatively for HCN-1A under basal conditions.
30 Ginkgo biloba and donepezil: a comparison in the treatment of Alzheimer's dementia in a randomized placebo-controlled double-blind study.
Mazza M, Capuano A, Bria P, Mazza S.
Eur J Neurol. 2006 Sep;13(9):981-5.
Department of Psychiatry, Catholic University of Sacred Heart, Rome, Italy. mariannamazza@hotmail.com
The Ginkgo biloba special extract EGb 761 seems to produce neuroprotective effects in neurodegenerative diseases of multifactorial origin. There is still debate about the efficacy of Ginkgo biloba special extract EGb 761 compared with second-generation cholinesterase inhibitors in the treatment of mild to moderate Alzheimer's dementia. Our aim is to assess the efficacy of the Ginkgo biloba special extract E.S. in patients with dementia of the Alzheimer type in slowing down the disease's degenerative progression and the patients' cognitive impairment compared with donepezil and placebo. The trial was designed as a 24-week randomized, placebo-controlled, double-blind study. Patients aged 50-80 years, suffering from mild to moderate dementia, were allocated into one of the three treatments: Ginkgo biloba (160 mg daily dose), donepezil (5 mg daily dose), or placebo group. The degree of severity of dementia was assessed by the Syndrom Kurz test and the Mini-Mental State Examination. Clinical Global Impression score was recorded to assess the change in the patients' conditions and the therapeutic efficacy of tested medications. Our results confirm the clinical efficacy of Ginkgo biloba E.S. (Flavogin) in the dementia of the Alzheimer type, comparable with donepezil clinical efficacy. There are few published trials that have directly compared a cholinesterase inhibitor with Ginkgo for dementia. This study directly compares a cholinesterase inhibitor with Ginkgo biloba for dementia of the Alzheimer type and could be a valid contribution in this debate. Our study suggests that there is no evidence of relevant differences in the efficacy of EGb 761 and donepezil in the treatment of mild to moderate Alzheimer's dementia, so the use of both substances can be justified. In addition, this study contributes to establish the efficacy and tolerability of the Ginkgo biloba special extract E.S. in the dementia of the Alzheimer type with special respect to moderately severe stages.
31 [Delay in progression of dependency and need of care of dementia patients treated with Ginkgo special extract EGb 761]
Wien Med Wochenschr. 2004 Nov;154(21-22):511-4.
In studies on the efficacy of antidementia drugs, a delay in symptom progression was often postulated based on a comparison of the change upon treatment and an assumed "natural" progression. Such comparisons were usually based on the cognitive subscore of the Alzheimer's Disease Assessment Scale (ADAS-cog), using either the drug-placebo differences after randomized treatment or the changes upon active drug treatment in open-label extension studies. Considering quality of life, competence, cost of care, and economics of therapeutic measures, a delay in the progression of dependency and need of care appears to be more relevant than a delay in cognitive abilities not directly related to activities of daily living. Therefore, for dementia patients treated with the Ginkgo special extract EGb 761, the delay in loss of capacities needed to cope with the demands of daily living was estimated, based on the Geriatric Evaluation of Relative's Rating Instrument (GERRI). The drug-placebo differences documented after 26 and 52 weeks of treatment corresponded to a delay in progression by 10 and 21 months, respectively. Regarding the subgroup with dementia of the Alzheimer type, the estimated delay was 16 and 25 months, respectively. It could thus be shown that by treatment with EGb 761 the progression of dependency and need of care can be slowed down, which may have an impact on costs for care, e.g. by delaying nursing home placement.
32 [Effect of Coenzyme Q10 and Ginkgo biloba on warfarin dosage in patients on long-term warfarin treatment. A randomized, double-blind, placebo-controlled cross-over trial]
Engelsen J, Nielsen JD, Hansen KF.
Ugeskr Laeger. 2003 Apr 28;165(18):1868-71.
[Article in Danish]
Klinisk Biokemisk Afdeling, Koagulationslaboratoriet, Amtssygehuset i Gentofte, Niels Andersens Vej 165, DK-2900 Hellerup. jeng@dadlnet.dk
INTRODUCTION: A few case-stories claim that the anti-oxidant Coenzyme Q10 and possibly also Ginkgo biloba interact with warfarin treatment. A decreased response to warfarin in the Coenzyme Q10 cases and an increased response in the Ginkgo biloba case have been described. MATERIAL AND METHODS: Twenty-four outpatients on stable, long-term warfarin treatment were included in a randomised, double blind, placebo-controlled crossover trial. Coenzyme Q10 100 mg daily, Ginkgo-Biloba 100 mg daily and placebo were given in random order over treatment periods of four weeks, each followed by a two week wash out period. The international normalized ratio (INR) INR was kept between 2.0 and 4.0 by appropriate adjustment of the warfarin dosage. RESULTS: Fourteen women and ten men, median ages 64.5 years (33-79) were included. Three patients withdrew from the study for personal reasons. The INR was stable during all treatment periods. The geometric mean dosage of warfarin did not change during the treatment periods: Ginkgo biloba 36.7 mg/week (95% confidence interval: 29.2-46.0); CoQ10 36.5 mg/week (29.1-45.8); placebo 36.0 mg/week (28.6-45.1). CONCLUSION: The study indicated that Coenzyme Q10 and Ginkgo biloba do not influence the clinical effect of warfarin.
33 Fatty acid intake and the risk of dementia and cognitive decline: a review of clinical and epidemiological studies. J Nutr Health Aging 2000;4(4):202-7
Dietary intake of fatty acids may be related to dementia and cognitive function through a number of plausible mechanisms, such as atherosclerosis and thrombosis, inflammation, via an effect on brain development and membrane functioning, or via accumulation of beta-amyloid. This review gives an overview of the few studies that have investigated the relationship between fatty acid intake (including the fatty acids from fish) and cognitive function or dementia and summarises the results from two Dutch population-based prospective studies: the Zutphen Elderly Study (n=476) and the Rotterdam Study (n=5,386). Additionally, limitations on dietary intake studies are discussed and possible mechanisms behind the investigated associations. Data from the Rotterdam Study showed that high intakes of the following nutrients were associated with an increased risk of dementia after adjustment for confounders: total fat (RR=2.4 (95%CI: 1.1-5.2)), saturated fat (RR=1.9 (95%CI: 0.9-4.0)), and cholesterol (RR=1.7 (95%CI: 0.9-3.2)). A high fish consumption, an important source of n-3 PUFAs, reduced the risk of dementia (RR=0.4 (95%CI: 0.2-0.9)). In the Zutphen Elderly Study a high linoleic acid intake was associated with cognitive impairment (OR=1.8 (95%CI: 1.0-3.0)). A high fish consumption tended to be inversely associated with cognitive impairment and decline (RR=0.5, 95%CI: 0.2-1.2). Since diet is a risk factor that is suitable for intervention these results are hopeful and potentially very important.
34 Dietary fat intake and the risk of incident dementia in the Rotterdam Study. Ann Neurol 1997 Nov;42(5):776-82
A high intake of saturated fat and cholesterol and a low intake of polyunsaturated fatty acids have been related to an increased risk of cardiovascular disease. Cardiovascular disease has been associated with dementia. We investigated the association between fat intake and incident dementia among participants, age 55 years or older, from the population-based prospective Rotterdam Study. Food intake of 5,386 nondemented participants was assessed at baseline with a semiquantitative food-frequency questionnaire. At baseline and after an average of 2.1 years of follow-up, we screened for dementia with a three-step protocol that included a clinical examination. The risk of dementia at follow-up (RR [95% CI]) was assessed with logistic regression. After adjustment for age, sex, education, and energy intake, high intakes of the following nutrients were associated with an increased risk of dementia: total fat (RR = 2.4 [1.1-5.2]), saturated fat (RR = 1.9 [0.9-4.0]), and cholesterol (RR = 1.7 [0.9-3.2]). Dementia with a vascular component was most strongly related to total fat and saturated fat. Fish consumption, an important source of n-3 polyunsaturated fatty acids, was inversely related to incident dementia (RR = 0.4 [0.2-0.91), and in particular to Alzheimer's disease (RR = 0.3 [0.1-0.9]). This study suggests that a high saturated fat and cholesterol intake increases the risk of dementia, whereas fish consumption may decrease this risk.
35 Diet and risk of dementia: Does fat matter?: The Rotterdam Study. Neurology 2002 Dec 24;59(12):1915-21
OBJECTIVE: To examine whether high intake of total fat, saturated fatty acids (saturated fat), trans fatty acids (trans fat), and cholesterol and low intake of monounsaturated fatty acids (MUFA), polyunsaturated fatty acids (PUFA), n-6 PUFA, and n-3 PUFA are associated with increased risk of dementia and its subtypes. METHOD: Data from the Rotterdam Study, a prospective cohort study among elderly, were used. At baseline (1990 to 1993), 5,395 subjects had normal cognition, were noninstitutionalized, and underwent complete dietary assessment by a semiquantitative food-frequency questionnaire. The cohort was continuously monitored for incident dementia, and re-examinations were performed in 1993 to 1994 and 1997 to 1999. The association between fat intake and incident dementia was examined by Cox's proportional hazards models. RESULTS: After a mean follow-up of 6.0 years, 197 subjects developed dementia (146 AD, 29 vascular dementia). High intake of total, saturated, trans fat, and cholesterol and low intake of MUFA, PUFA, n-6 PUFA, and n-3 PUFA were not associated with increased risk of dementia or its subtypes. Rate ratios of dementia per standard deviation increase in intake were for total fat 0.93 (95% CI 0.81 to 1.07), for saturated fat 0.91 (95% CI 0.79 to 1.05), for trans fat 0.90 (95% CI 0.77 to 1.06), for cholesterol 0.93 (95% CI 0.80 to 1.08), for MUFA 0.96 (95% CI 0.84 to 1.10), for PUFA 1.05 (95% CI 0.80 to 1.38), for n-6 PUFA 1.03 (95% CI 0.77 to 1.36), and for n-3 PUFA 1.07 (95% CI 0.94 to 1.22). CONCLUSION: High intake of total, saturated, and trans fat and cholesterol and low intake of MUFA, PUFA, n-6 PUFA, and n-3 PUFA were not associated with increased risk of dementia or its subtypes.
36 Potential role of dietary n-3 fatty acids in the prevention of dementia and macular degeneration. Am J Clin Nutr 2006 Jun;83(6 Suppl):1494S-1498S
Dementia and age-related macular degeneration (AMD) are major causes of disability in the elderly. n-3 Fatty acids, particularly docosahexaenoic acid (DHA), are highly concentrated in brain and retinal tissue and may prevent or delay the progression of dementia and AMD. Low dietary intakes and plasma concentrations have been reported to be associated with dementia, cognitive decline, and AMD risk. The major dietary sources of DHA are fish and fish oils, although dietary supplements are available. At this point, it is not possible to make firm recommendations regarding n-3 fatty acids and the prevention of dementia and AMD. Our own unpublished observations from the Framingham Heart Study suggest that > or =180 mg/d of dietary DHA (approximately 2.7 fish servings/wk) is associated with an approximately 50% reduction in dementia risk. At least this amount of DHA is generally found in one commercially available 1-g fish oil capsule given daily.
37 Arch Neurol 2003 Jul;60(7):940-6
38 The National Library of Medicine contains some smaller studies that showed no apparent benefit from fish oils, but most of these lasted only two months or so. This tells me only that you can't make a material difference in our three pound, fat-rich brain without a longer intervention.
39 Dietary fat intake and 6-year cognitive change in an older biracial community population. Neurology 2004 May 11;62(9):1573-9
OBJECTIVE: To examine whether consumption of different types of fat is associated with age-related change in cognition. METHODS: The authors related fat consumption to 6-year change in cognitive function among 2,560 participants of the Chicago Health and Aging Project, ages 65 and older, with no history of heart attack, stroke, or diabetes at baseline. Fat intake was measured by food frequency questionnaire. Cognitive function was measured at baseline and 3-year and 6-year follow-ups, using the average z score of four cognitive tests: the East Boston Tests of Immediate and Delayed Recall, the Mini-Mental State Examination, and the Symbol Digit Modalities Test. RESULTS: In separate mixed models adjusted for demographic and cardiovascular risk factors and intakes of antioxidant nutrients and other dietary fats, higher intakes of saturated fat (p for trend = 0.04) and trans-unsaturated fat (p for trend = 0.07) were linearly associated with greater decline in cognitive score over 6 years. These associations became stronger in analyses that eliminated persons whose fat intake changed in recent years or whose baseline cognitive scores were in the lowest 15%. Inverse associations with cognitive decline were observed in these latter restricted analyses for high intake of monounsaturated fat and a high ratio of polyunsaturated to saturated fat intake. Intakes of total fat, vegetable and animal fats, and cholesterol were not associated with cognitive change. CONCLUSION: A diet high in saturated or trans-unsaturated fat or low in nonhydrogenated unsaturated fats may be associated with cognitive decline among older persons.
40 Dietary intake of fatty acids and fish in relation to cognitive performance at middle age. Neurology 2004 Jan 27;62(2):275-80
OBJECTIVE: To examine the associations of fatty acid and fish intake with cognitive function. METHODS: Data are from a cross-sectional population-based study among 1,613 subjects ranging from 45 to 70 years old. From 1995 until 2000, an extensive cognitive battery was administered and compound scores were constructed for memory, psychomotor speed, cognitive flexibility (i.e., higher order information processing), and overall cognition. A self-administered food-frequency questionnaire was used to assess habitual food consumption. The risk of impaired cognitive function (lowest 10% of the compound score) according to the energy adjusted intake of fatty acids was assessed with logistic regression, adjusting for age, sex, education, smoking, alcohol consumption, and energy intake. RESULTS: Marine omega-3 polyunsaturated fatty acids (PUFA) (eicosapentaenoic acid and docosahexaenoic acid) were inversely related to the risk of impaired overall cognitive function and speed (per SD increase: OR = 0.81, 95% CI 0.66 to 1.00 and OR = 0.72, 95% CI 0.57 to 0.90). Results for fatty fish consumption were similarly inverse. Higher dietary cholesterol intake was significantly associated with an increased risk of impaired memory and flexibility (per SD increase: OR = 1.27, 95% CI 1.02 to 1.57 and OR = 1.26, 95% CI 1.01 to 1.57). Per SD increase in saturated fat intake, the risk of impaired memory, speed, and flexibility was also increased, although not significantly. CONCLUSIONS: Fatty fish and marine omega-3 PUFA consumption was associated with a reduced risk and intake of cholesterol and saturated fat with an increased risk of impaired cognitive function in this middle-aged population.
41 Roles of unsaturated fatty acids (especially omega-3 fatty acids) in the brain at various ages and during ageing. J Nutr Health Aging 2004;8(3):163-74
Among various organs, in the brain, the fatty acids most extensively studied are omega-3 fatty acids. Alpha-linolenic acid (18:3omega3) deficiency alters the structure and function of membranes and induces minor cerebral dysfunctions, as demonstrated in animal models and subsequently in human infants. Even though the brain is materially an organ like any other, that is to say elaborated from substances present in the diet (sometimes exclusively), for long it was not accepted that food can have an influence on brain structure, and thus on its function. Lipids, and especially omega-3 fatty acids, provided the first coherent experimental demonstration of the effect of diet (nutrients) on the structure and function of the brain. In fact the brain, after adipose tissue, is the organ richest in lipids, whose only role is to participate in membrane structure. First it was shown that the differentiation and functioning of cultured brain cells requires not only alpha-linolenic acid (the major component of the omega-3, omega3 family), but also the very long omega-3 and omega-6 carbon chains (1). It was then demonstrated that alpha-linolenic acid deficiency alters the course of brain development, perturbs the composition and physicochemical properties of brain cell membranes, neurones, oligodendrocytes, and astrocytes (2).This leads to physicochemical modifications, induces biochemical and physiological perturbations, and results in neurosensory and behavioural upset (3). Consequently, the nature of polyunsaturated fatty acids (in particular omega-3) present in formula milks for infants (premature and term) conditions the visual and cerebral abilities, including intellectual. Moreover, dietary omega-3 fatty acids are certainly involved in the prevention of some aspects of cardiovascular disease (including at the level of cerebral vascularization), and in some neuropsychiatric disorders, particularly depression, as well as in dementia, notably Alzheimer's disease. Recent results have shown that dietary alpha-linolenic acid deficiency induces more marked abnormalities in certain cerebral structures than in others, as the frontal cortex and pituitary gland are more severely affected. These selective lesions are accompanied by behavioural disorders more particularly affecting certain tests (habituation, adaptation to new situations). Biochemical and behavioural abnormalities are partially reversed by a dietary phospholipid supplement, especially omega-3-rich egg yolk extracts or pig brain. A dose-effect study showed that animal phospholipids are more effective than plant phospholipids to reverse the consequences of alpha-linolenic acid deficiency, partly because they provide very long preformed chains. Alpha-linolenic acid deficiency decreases the perception of pleasure, by slightly altering the efficacy of sensory organs and by affecting certain cerebral structures. Age-related impairment of hearing, vision and smell is due to both decreased efficacy of the parts of the brain concerned and disorders of sensory receptors, particularly of the inner ear or retina. For example, a given level of perception of a sweet taste requires a larger quantity of sugar in subjects with alpha-linolenic acid deficiency. In view of occidental eating habits, as omega-6 fatty acid deficiency has never been observed, its impact on the brain has not been studied. In contrast, omega-9 fatty acid deficiency, specifically oleic acid deficiency, induces a reduction of this fatty acid in many tissues, except the brain (but the sciatic nerve is affected). This fatty acid is therefore not synthesized in sufficient quantities, at least during pregnancy-lactation, implying a need for dietary intake. It must be remembered that organization of the neurons is almost complete several weeks before birth, and that these neurons remain for the subject's life time. Consequently, any disturbance of these neurons, an alteration of their connections, and impaired turnover of their constituents at any stage of life, will tend to accelerate ageing. The enzymatic activities of sytivities of synthesis of long-chain polyunsaturated fatty acids from linoleic and alpha-linolenic acids are very limited in the brain: this organ therefore depends on an exogenous supply. Consequently, fatty acids that are essential for the brain are arachidonic acid and cervonic acid, derived from the diet, unless they are synthesized by the liver from linoleic acid and alpha-linolenic acid. The age-related reduction of hepatic desaturase activities (which participate in the synthesis of long chains, together with elongases) can impair turnover of cerebral membranes. In many structures, especially in the frontal cortex, a reduction of cervonic and arachidonic acids is observed during ageing, predominantly associated with a reduction of phosphatidylethanolamines (mainly in the form of plasmalogens). Peroxisomal oxidation of polyunsaturated fatty acids decreases in the brain during ageing, participating in decreased turnover of membrane fatty acids, which are also less effectively protected against peroxidation by free radicals.
42 Fatty acid analysis of blood plasma of patients with Alzheimer's disease, other types of dementia, and cognitive impairment. Lipids 2000 Dec;35(12):1305-12
Fatty acid differences, including docosahexaenoic acid (DHA; 22:6n-3) have been shown in the brains of Alzheimer's patients (AD) as compared with normal age-matched individuals. Furthermore, low serum DHA is a significant risk factor for the development of AD. The relative concentration of DHA and other fatty acids, however, in the plasma of AD patients compared with patients with other kinds of dementias (other dementias; OD), patients who are cognitively impaired but nondemented (CIND), or normal patients is not known. In this study we analyzed the total phospholipid, phosphatidylcholine (PC), phosphatidylethanolamine (PE), and lysophosphatidylcholine (lysoPC) fractions of plasma from patients diagnosed with AD, OD, or CIND and compared them with a group of elderly control subjects with normal cognitive functioning. Plasma phospholipid and PC levels of 20:5n-3, DHA, total n-3 fatty acids, and the n-3/n-6 ratio were lower in the AD, OD, and CIND groups. Plasma phospholipid 24:0 was lower in the AD, OD, and CIND groups as compared with the group of control patients, and total n-6 fatty acid levels were higher in the AD and CIND groups only. In the plasma PE fraction, levels of 20:5n-3, DHA, and the total n-3 fatty acid levels were significantly lower in the AD, OD, and CIND groups. DHA levels were lower in the lysoPC fraction of CIND individuals only. There were no other differences in the fatty acid compositions of the different phospholipid fractions. Therefore, in AD, OD, and CIND individuals, low levels of n-3 fatty acids in the plasma may be a risk factor for cognitive impairment and/or dementia. Interestingly, a decreased level of plasma DHA was not limited to the AD patients but appears to be common in cognitive impairment with aging.
43 Dietary fat intake and 6-year cognitive change in an older biracial community population. Neurology 2004 May 11;62(9):1573-9
Rush Institute for Healthy Aging, Rush University Medical Center, Chicago, IL 60612, USA.
OBJECTIVE: To examine whether consumption of different types of fat is associated with age-related change in cognition. METHODS: The authors related fat consumption to 6-year change in cognitive function among 2,560 participants of the Chicago Health and Aging Project, ages 65 and older, with no history of heart attack, stroke, or diabetes at baseline. Fat intake was measured by food frequency questionnaire. Cognitive function was measured at baseline and 3-year and 6-year follow-ups, using the average z score of four cognitive tests: the East Boston Tests of Immediate and Delayed Recall, the Mini-Mental State Examination, and the Symbol Digit Modalities Test. RESULTS: In separate mixed models adjusted for demographic and cardiovascular risk factors and intakes of antioxidant nutrients and other dietary fats, higher intakes of saturated fat (p for trend = 0.04) and trans-unsaturated fat (p for trend = 0.07) were linearly associated with greater decline in cognitive score over 6 years. These associations became stronger in analyses that eliminated persons whose fat intake changed in recent years or whose baseline cognitive scores were in the lowest 15%. Inverse associations with cognitive decline were observed in these latter restricted analyses for high intake of monounsaturated fat and a high ratio of polyunsaturated to saturated fat intake. Intakes of total fat, vegetable and animal fats, and cholesterol were not associated with cognitive change. CONCLUSION: A diet high in saturated or trans-unsaturated fat or low in nonhydrogenated unsaturated fats may be associated with cognitive decline among older persons.
44 A review of magnesium intake in the elderly. A cause for concern? Elderly people in the United States represent an emerging high risk group for nutritional deficiencies. A magnesium deficit in the elderly can occur due to inadequate nutrient intakes, multiple drug use, or altered gastrointestinal function. Magnesium has been targeted as a risk factor for elderly people and has been implicated in the aging process. Data presented in this review confirm decreased availability of magnesium in the food supply, lower intakes of magnesium by elderly people, and widespread supplementation practices. Conflicting data exist regarding levels of magnesium in the blood and magnesium status in the elderly. It is not known to what extent suboptimal intakes may affect the aging process; however, magnesium-deficient conditions have been associated with neuromuscular and cardiovascular disorders, endocrine disturbances, insulin resistance and Alzheimer's disease.
Magnes Res 1992 Mar;5(1):61-7